Welcome to China Oncology,
China Oncology ›› 2024, Vol. 34 ›› Issue (2): 176-184.doi: 10.19401/j.cnki.1007-3639.2024.02.005
• Article • Previous Articles Next Articles
CHEN Yuanxiang( ), YU Tao, YANG Shiyu, ZENG Tao, WEI Lan, ZHANG Yan(
), YU Tao, YANG Shiyu, ZENG Tao, WEI Lan, ZHANG Yan( )
)
Received:2023-09-19
Revised:2023-12-18
Online:2024-02-29
Published:2024-03-14
Contact:
ZHANG Yan
Share article
CLC Number:
CHEN Yuanxiang, YU Tao, YANG Shiyu, ZENG Tao, WEI Lan, ZHANG Yan. KDM4A promotes the migration and invasion of breast cancer cell line MDA-MB-231 by downregulating BMP9[J]. China Oncology, 2024, 34(2): 176-184.
Tab. 1
Sequences of siRNA and RTFQ-PCR primer"
| Primer | Sequence (5'→3') |
|---|---|
| siRNA negative control | F: UUCUCCGAACGUGUCACGUTT |
| R: ACGUGACACGUUCGGAGAATT | |
| siKDM4A | F: CCGAGUUUGUCUUGAAAUATT |
| R: UAUUUCAAGACAAACUCGGTT | |
| BMP9 | F: CGTCCAACATTGTGCGGAG |
| R: GACAGGAGACATAGAGTCGGAG | |
| KDM4A | F: CCTCACTGCGCTGTCTGTAT |
| R: CCAGTCGAAGTGAAGCACAT | |
| GAPDH | F: CAGCGACACCCACTCCTC |
| R: TGAGGTCCACCACCCTGT |
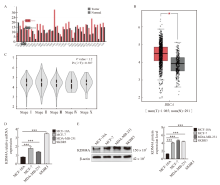
Fig. 1
KDM4A highly expressed in breast cancer A, B: High expression of KDM4A in breast cancer; C: Expression of KDM4A in different stages of breast cancer; D: The expression of KDM4A in breast cancer cells were detected by RTFQ-PCR; E: The expression of KDM4A in breast cancer cells was detected by Western blot. *: P<0.05, tumor vs normal; ***: P<0.001, compared with MCF-10A."
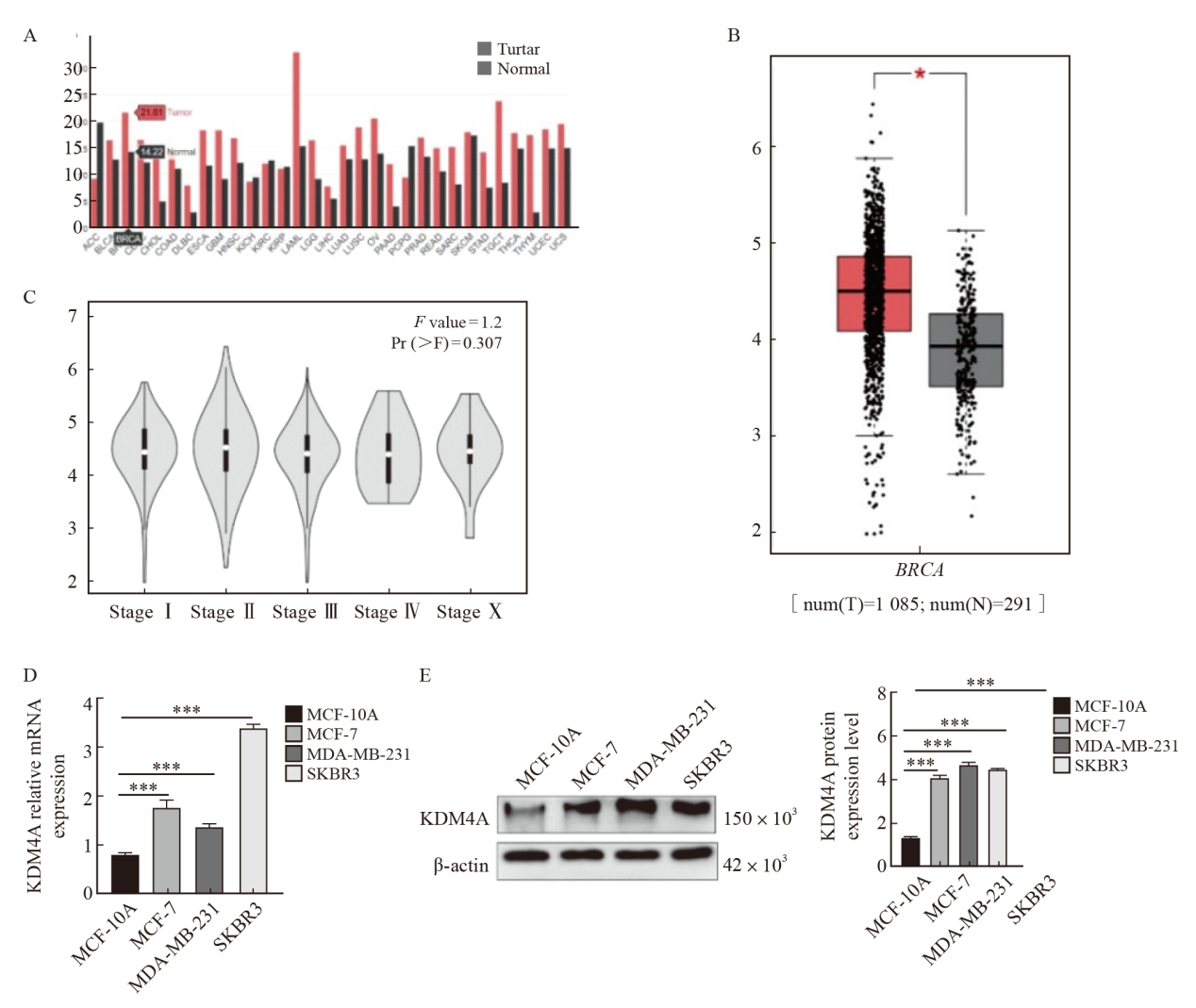
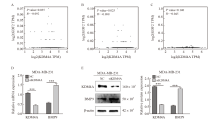
Fig. 2
The expression of KDM4A is negatively correlated with BMP9 A-C: The expression of KDM4A and BMP9 in HNSC, KIRC and BRCA; D: Knocking down KDM4A significantly increased the mRNA expression of BMP9 in MDA-MB-231; E: Knocking down KDM4A significantly increased the protein expression of BMP9 in MDA-MB-231. ***: P<0.001, compared with NC group. NC: Normal control."
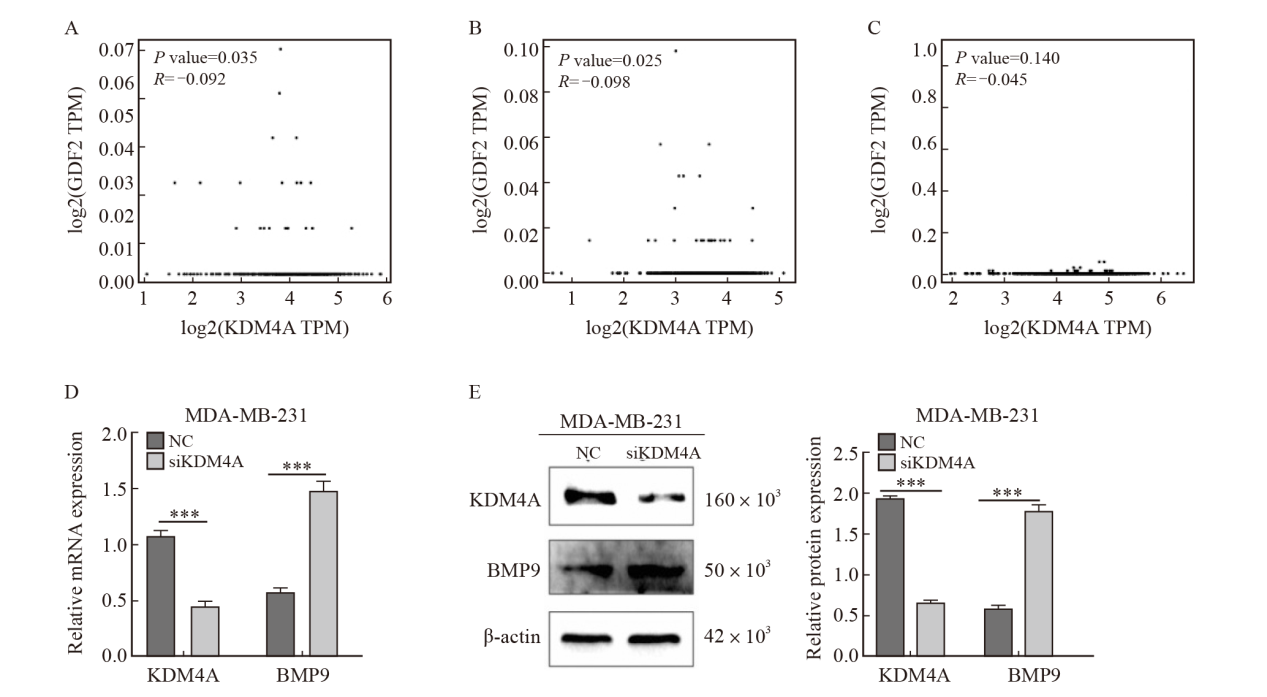

Fig. 3
KDM4A downregulatd histone methylation status A: Knocking down KDM4A upregulates H3K36 and H3K4 methylation levels; B: ChIP detection of H3K36me3 and H3K4me3 enrichment in the BMP9 promoter region in MDA-MB-231 cells. ***: P<0.001, compared with NC group. NC: Normal control; ns: No significance."
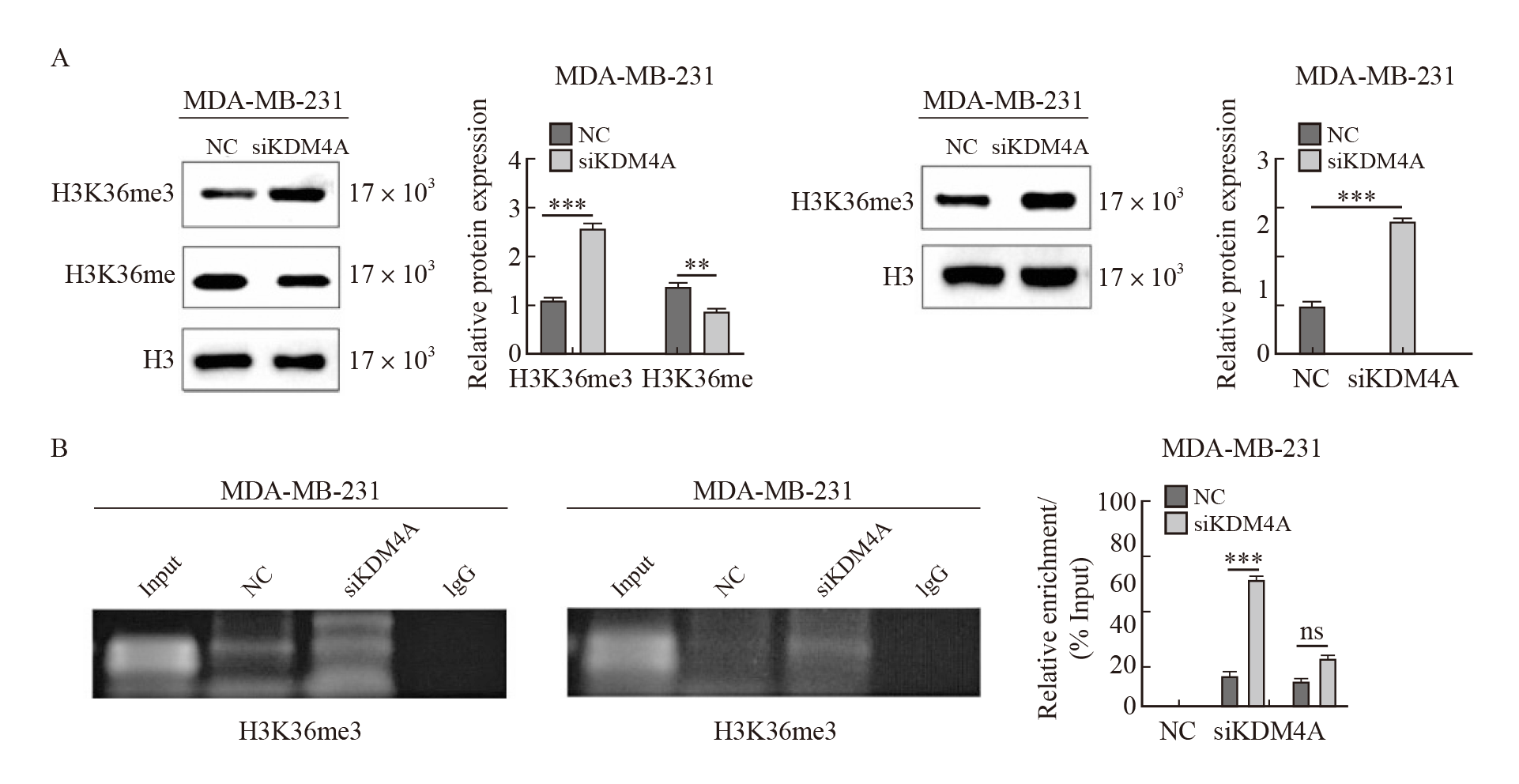
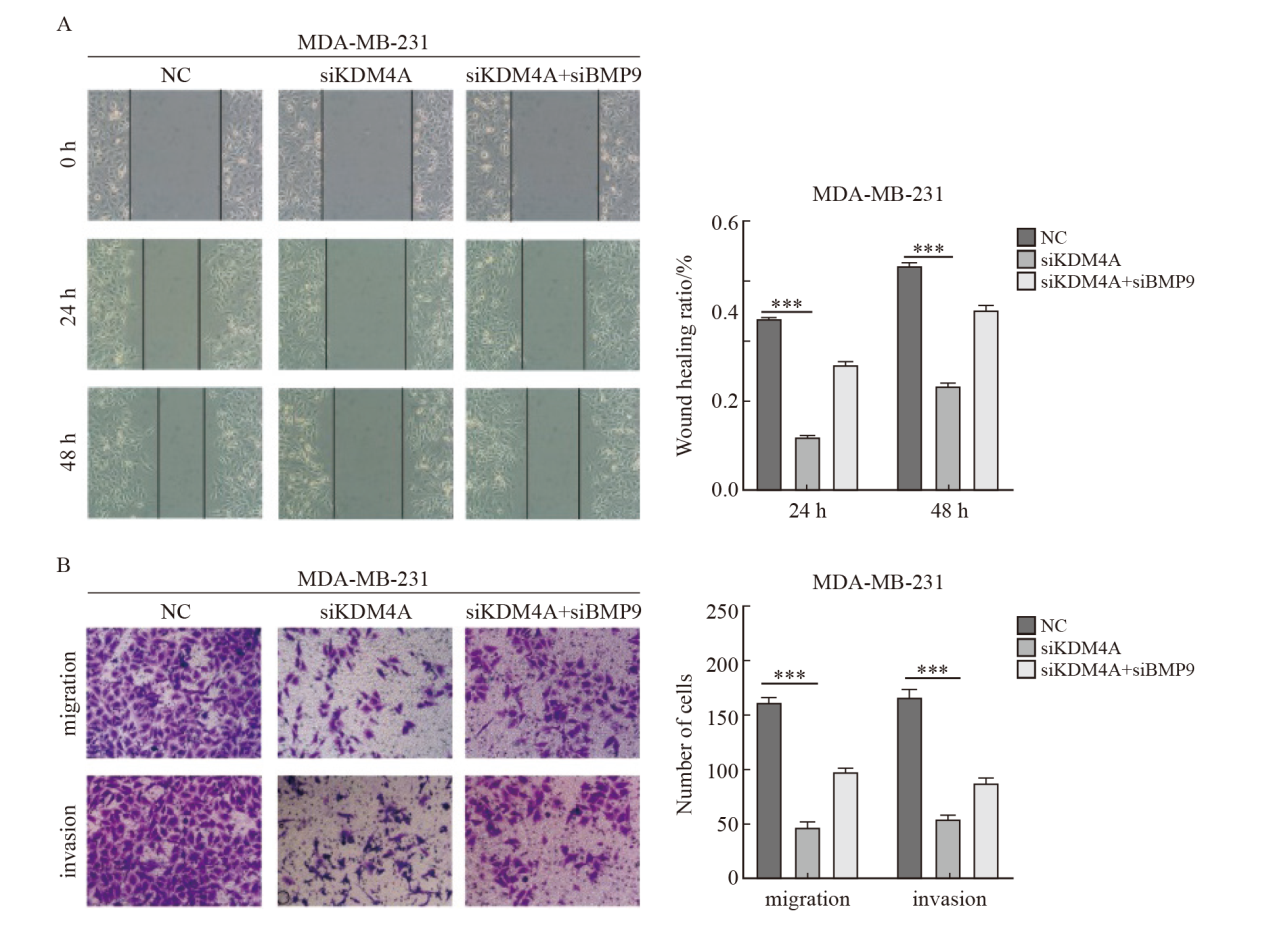
Fig.5
Silencing of KDM4A inhibited the migration and invasion of breast cancer cells A: The migration ability of MDA-MB-231 was detected by wound healing test; B: The migration and invasion ability of MDA-MB-231 was detected by transwell assay. ***: P<0.001, compared with NC group. NC: Normal control."
| [1] |
HOUGHTON S C, HANKINSON S E. Cancer progress and priorities: breast cancer[J]. Cancer Epidemiol Biomarkers Prev, 2021, 30(5): 822-844.
doi: 10.1158/1055-9965.EPI-20-1193 |
| [2] |
BARZAMAN K, KARAMI J, ZAREI Z, et al. Breast cancer: biology, biomarkers, and treatments[J]. Int Immunopharmacol, 2020, 84: 106535.
doi: 10.1016/j.intimp.2020.106535 |
| [3] | LANDSKRON G, DE LA FUENTE M, THUWAJIT P, et al. Chronic inflammation and cytokines in the tumor microenvironment[J]. J Immunol Res, 2014, 2014: 149185. |
| [4] |
KONG R N, GAO J, JI L M, et al. Iguratimod ameliorates rheumatoid arthritis progression through regulating miR-146a mediated IRAK1 expression and TRAF6/JNK1 pathway: an in vivo and in vitro study[J]. Clin Exp Rheumatol, 2021, 39(2): 289-303.
doi: 10.55563/clinexprheumatol/urhbn0 |
| [5] | 谭秋芬, 胡惠军. FEN1、GTF2IP23、KDM4A在乳腺癌组织中的表达研究[J]. 国际检验医学杂志, 2023, 44(3): 311-315. |
| TAN Q F, HU H J. Expression and correlation of FEN1, GTF2IP23, and KDM4A in breast cancer tissues[J]. Int J Lab Med, 2023, 44(3): 311-315. | |
| [6] |
LEE J, THOMPSON J R, BOTUYAN M V, et al. Distinct binding modes specify the recognition of methylated histones H3K4 and H4K20 by JMJD2A-tudor[J]. Nat Struct Mol Biol, 2008, 15(1): 109-111.
doi: 10.1038/nsmb1326 pmid: 18084306 |
| [7] | CHEN Y C, LIU X R, LI Y K, et al. Lung cancer therapy targeting histone methylation: opportunities and challenges[J]. Comput Struct Biotechnol J, 2018, 16: 211-223. |
| [8] |
WANG G L, WEN B Q, DENG Z C, et al. Endothelial progenitor cells stimulate neonatal lung angiogenesis through FOXF1-mediated activation of BMP9/ACVRL1 signaling[J]. Nat Commun, 2022, 13(1): 2080.
doi: 10.1038/s41467-022-29746-y pmid: 35440116 |
| [9] |
XU J Z, ZHOU Y M, ZHANG L L, et al. BMP9 reduces age-related bone loss in mice by inhibiting osteoblast senescence through Smad1-Stat1-P21 axis[J]. Cell Death Discov, 2022, 8(1): 254.
doi: 10.1038/s41420-022-01048-8 |
| [10] |
HAN Y L, PAN Q Z, GUO Z X, et al. BMP9-induced vascular normalisation improves the efficacy of immunotherapy against hepatitis B virus-associated hepatocellular carcinoma[J]. Clin Transl Med, 2023, 13(5): e1247.
doi: 10.1002/ctm2.1247 pmid: 37132170 |
| [11] |
CHEN H, NIO K, TANG H, et al. BMP9-ID1 signaling activates HIF-1α and VEGFA expression to promote tumor angiogenesis in hepatocellular carcinoma[J]. Int J Mol Sci, 2022, 23(3): 1475.
doi: 10.3390/ijms23031475 |
| [12] |
YU H M, CHEN Y X, LANG L, et al. BMP9 promotes autophagy and inhibits migration and invasion in breast cancer cells through the c-Myc/SNHG3/mTOR signaling axis[J]. Tissue Cell, 2023, 82: 102073.
doi: 10.1016/j.tice.2023.102073 |
| [13] |
WANG W, WENG Y G, REN W, et al. Biological roles of human bone morphogenetic protein 9 in the bone microenvironment of human breast cancer MDA-MB-231 cells[J]. Am J Transl Res, 2015, 7(9): 1660-1674.
pmid: 26550465 |
| [14] |
WANG T, ZHANG Z H, WANG K, et al. Inhibitory effects of BMP9 on breast cancer cells by regulating their interaction with pre-adipocytes/adipocytes[J]. Oncotarget, 2017, 8(22): 35890-35901.
doi: 10.18632/oncotarget.16271 pmid: 28415788 |
| [15] |
REN W, SUN X X, WANG K, et al. BMP9 inhibits the bone metastasis of breast cancer cells by downregulating CCN2 (connective tissue growth factor, CTGF) expression[J]. Mol Biol Rep, 2014, 41(3): 1373-1383.
doi: 10.1007/s11033-013-2982-8 pmid: 24413988 |
| [16] |
LI S, DAI H Y, HE Y, et al. BMP9 inhibits the growth of breast cancer cells by downregulation of the PI3K/Akt signaling pathway[J]. Oncol Rep, 2018, 40(3): 1743-1751.
doi: 10.3892/or.2018.6572 pmid: 30015950 |
| [17] |
OUARNÉ M, BOUVARD C, BONEVA G, et al. BMP9, but not BMP10, acts as a quiescence factor on tumor growth, vessel normalization and metastasis in a mouse model of breast cancer[J]. J Exp Clin Cancer Res, 2018, 37(1): 209.
doi: 10.1186/s13046-018-0885-1 pmid: 30165893 |
| [18] |
NEVES REBELLO ALVES L, DUMMER MEIRA D, POPPE MERIGUETI L, et al. Biomarkers in breast cancer: An old story with a new end[J]. Genes, 2023, 14(7): 1364.
doi: 10.3390/genes14071364 |
| [19] |
RANDO O J, CHANG H Y. Genome-wide views of chromatin structure[J]. Annu Rev Biochem, 2009, 78: 245-271.
doi: 10.1146/annurev.biochem.78.071107.134639 pmid: 19317649 |
| [20] | WEI C, DENG X G, GAO S D, et al. Cantharidin inhibits proliferation of liver cancer by inducing DNA damage via KDM4A-dependent histone H3K36 demethylation[J]. Evid Based Complement Alternat Med, 2022, 2022: 2197071. |
| [21] |
SUN S S, YANG F J, ZHU Y C, et al. RETRACTED: KDM4A promotes the growth of non-small cell lung cancer by mediating the expression of Myc via DLX5 through the Wnt/β-catenin signaling pathway[J]. Life Sci, 2020, 262: 118508.
doi: 10.1016/j.lfs.2020.118508 |
| [22] |
CHEN M, JIANG Y H, SUN Y B. KDM4A-mediated histone demethylation of SLC7A11 inhibits cell ferroptosis in osteosarcoma[J]. Biochem Biophys Res Commun, 2021, 550: 77-83.
doi: 10.1016/j.bbrc.2021.02.137 |
| [23] | XIONG J, NIE M F, FU C, et al. Hypoxia enhances HIF1α transcription activity by upregulating KDM4A and mediating H3K9me3, thus inducing ferroptosis resistance in cervical cancer cells[J]. Stem Cells Int, 2022, 2022: 1608806. |
| [24] | LI Y, WANG Y N, XIE Z, et al. JMJD2A facilitates growth and inhibits apoptosis of cervical cancer cells by downregulating tumor suppressor miR-491-5p[J]. Mol Med Rep, 2019, 19(4): 2489-2496. |
| [25] |
LIN X, QIU W X, XIAO Y Y, et al. MiR-199b-5p suppresses tumor angiogenesis mediated by vascular endothelial cells in breast cancer by targeting ALK1[J]. Front Genet, 2019, 10: 1397.
doi: 10.3389/fgene.2019.01397 pmid: 32082362 |
| [26] |
REN W, LIU Y H, WAN S H, et al. BMP9 inhibits proliferation and metastasis of HER2-positive SK-BR-3 breast cancer cells through ERK1/2 and PI3K/AKT pathways[J]. PLoS One, 2014, 9(5): e96816.
doi: 10.1371/journal.pone.0096816 |
| [1] | XU Rui, WANG Zehao, WU Jiong. Advances in the role of tumor-associated neutrophils in the development of breast cancer [J]. China Oncology, 2024, 34(9): 881-889. |
| [2] | CAO Xiaoshan, YANG Beibei, CONG Binbin, LIU Hong. The progress of treatment for brain metastases of triple-negative breast cancer [J]. China Oncology, 2024, 34(8): 777-784. |
| [3] | ZHANG Jian. Clinical consideration of two key questions in assessing menopausal status of female breast cancer patients [J]. China Oncology, 2024, 34(7): 619-627. |
| [4] | JIANG Dan, SONG Guoqing, WANG Xiaodan. Study on the mechanism of mitochondrial dysfunction and CPT1A/ERK signal transduction pathway regulating malignant behavior in breast cancer [J]. China Oncology, 2024, 34(7): 650-658. |
| [5] | DONG Jianqiao, LI Kunyan, LI Jing, WANG Bin, WANG Yanhong, JIA Hongyan. A study on mechanism of SIRT3 inducing endocrine drug resistance in breast cancer via deacetylating YME1L1 [J]. China Oncology, 2024, 34(6): 537-547. |
| [6] | HAO Xian, HUANG Jianjun, YANG Wenxiu, LIU Jinting, ZHANG Junhong, LUO Yubei, LI Qing, WANG Dahong, GAO Yuwei, TAN Fuyun, BO Li, ZHENG Yu, WANG Rong, FENG Jianglong, LI Jing, ZHAO Chunhua, DOU Xiaowei. Establishment of primary breast cancer cell line as new model for drug screening and basic research [J]. China Oncology, 2024, 34(6): 561-570. |
| [7] | Committee of Breast Cancer Society, China Anti-Cancer Association. Expert consensus on clinical applications of ovarian function suppression for Chinese women with early breast cancer (2024 edition) [J]. China Oncology, 2024, 34(3): 316-333. |
| [8] | ZHANG Qi, XIU Bingqiu, WU Jiong. Progress of important clinical research of breast cancer in China in 2023 [J]. China Oncology, 2024, 34(2): 135-142. |
| [9] | ZHANG Siyuan, JIANG Zefei. Important research progress in clinical practice for advanced breast cancer in 2023 [J]. China Oncology, 2024, 34(2): 143-150. |
| [10] | WANG Zhaobu, LI Xing, YU Xinmiao, JIN Feng. Important research progress in clinical practice for early breast cancer in 2023 [J]. China Oncology, 2024, 34(2): 151-160. |
| [11] | LUO Yang, SUN Tao, SHAO Zhimin, CUI Jiuwei, PAN Yueyin, ZHANG Qingyuan, CHENG Ying, LI huiping, YANG Yan, YE Changsheng, YU Guohua, WANG Jingfen, LIU Yunjiang, LIU Xinlan, ZHOU Yuhong, BAI Yuju, GU Yuanting, WANG Xiaojia, XU Binghe, SONG Lihua. Efficacy, metabolic characteristics, safety and immunogenicity of AK-HER2 compared with reference trastuzumab in patients with metastatic HER2-positive breast cancer: a multicenter, randomized, double-blind phase Ⅲ equivalence trial [J]. China Oncology, 2024, 34(2): 161-175. |
| [12] | HU Xiaoyu, CAI Yuwen, YE Fugui, SHAO Zhimin, HU Weigang, YU Keda. Impact of BRCA1/2 germline mutation on the incidence of second primary cancer following postoperative radiotherapy in patients with triple-negative breast cancer [J]. China Oncology, 2024, 34(2): 185-190. |
| [13] | ZHANG Siwei, MA Ding, JIANG Yizhou, SHAO Zhimin. “Subtype-precise” therapy leads diagnostic and therapeutic innovations: a new pattern for precision treatment of breast cancer [J]. China Oncology, 2024, 34(11): 1045-1052. |
| [14] | OUYANG Fei, WANG Yang, CHEN Yu, PEI Guoqing, WANG Ling, ZHANG Yang, SHI Lei. Construction of the prediction model of breast cancer bone metastasis based on machine learning [J]. China Oncology, 2024, 34(10): 903-914. |
| [15] | ZHAO Junxiu, ZHU Yi, SONG Xiaoyu, ZHE Chao, XIAO Yuhan, LIU Yunduo, LI Linhai, XIAO Bin. Circ-0007766 acts as a miR-1972 sponge to promote breast cancer cell migration and invasion via upregulation of HER2 [J]. China Oncology, 2024, 34(10): 915-930. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
沪ICP备12009617
Powered by Beijing Magtech Co. Ltd
