Welcome to China Oncology,
China Oncology ›› 2023, Vol. 33 ›› Issue (5): 437-444.doi: 10.19401/j.cnki.1007-3639.2023.05.003
• Article • Previous Articles Next Articles
WANG Xuemei( ), CHENG Yu, QI Jiemin(
), CHENG Yu, QI Jiemin( )
)
Received:2022-07-22
Revised:2022-09-29
Online:2023-05-30
Published:2023-06-16
Contact:
QI Jiemin
Share article
CLC Number:
WANG Xuemei, CHENG Yu, QI Jiemin. PRMT7 inhibits proliferation and migration of bladder cancer cells by regulating Notch signaling pathway[J]. China Oncology, 2023, 33(5): 437-444.

Fig. 1
Expression of PRMT7 in bladder cancer cell lines Expression of total PRMT7 protein in bladder cancer cell lines 5637, T24, RT112, UM-UC-3 and ureteral epithelial immortalized cells SV-HUC-1. Compared with SV-HUC-1, total protein of PRMT7 was significantly higher in 5637 cells and significantly lower in T24 cells. *: P<0.05, compared with SV-HUC-1; **: P<0.01, compared with SV-HUC-1."
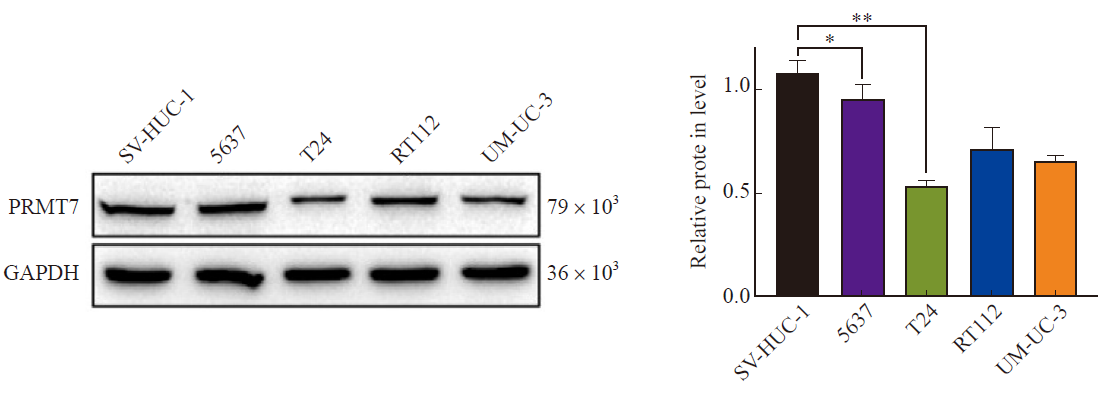
Fig. 2
Transfection efficiency of PRMT7 was verified Silenced and overexpressed PRMT7 protein and mRNA levels in bladder cancer 5637 and T24 cells. The results showed that the histone and mRNA expression levels of siPRMT7#1 and siPRMT7#2 were significantly decreased compared with si-NC, and the histone and mRNA expression levels of p-PRMT7 were significantly increased compared with p-PCMV3 group. **: P<0.01, compared with si-NC; ***: P<0.001, compared with si-NC or p-PCMV3."
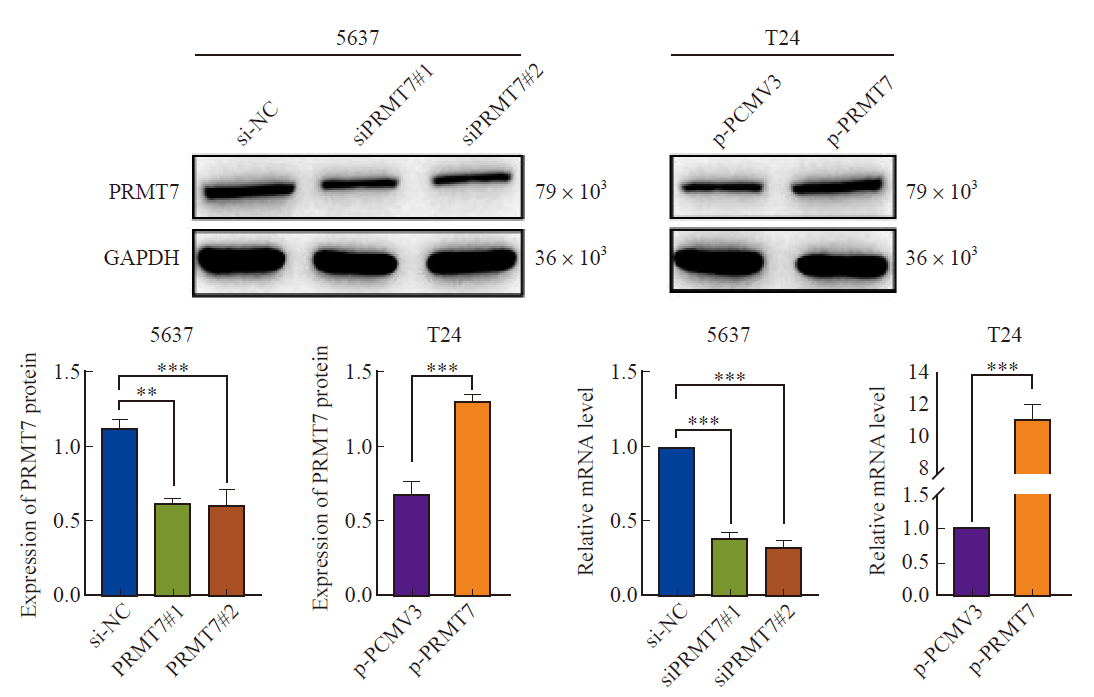
Fig. 3
PRMT7 inhibits the proliferation and migration of bladder cancer cells PRMT7 gene was silenced in 5637 cells, and PRMT7 gene was overexpressed in T24 cells. A: CCK-8 assay showed that compared with the control group, down-regulation of PRMT7 promoted cell growth, while overexpression of PRMT7 was opposite. B: Colony formation experiment showed that compared with the control group, down-regulation of PRMT7 promoted cell proliferation, while overexpression of PRMT7 was opposite. C: Transwell experiment showed that compared with the control group, down-regulation of PRMT7 promoted cell migration, while overexpression of PRMT7 was opposite. D: Western blot results showed that compared with the control group, the expression of Cyclin D1, CDK4 and MMP9 increased after down-regulation of PRMT7, while the overexpression of PRMT7 was on the contrary. *: P<0.05, compared with p-PCMV3; **: P<0.01, compared with si-NC or p-PCMV3; ***: P<0.001, compared with si-NC."
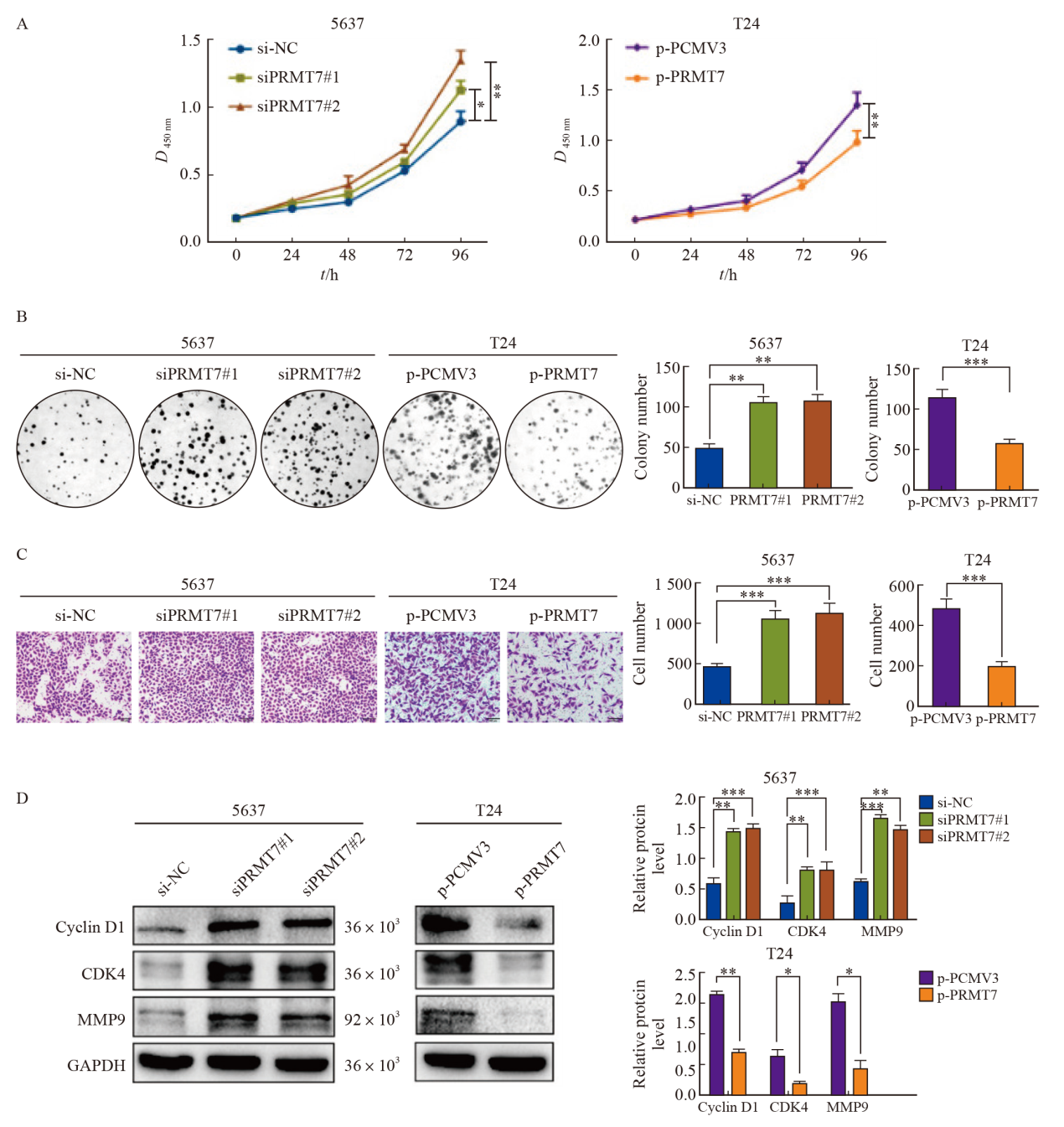
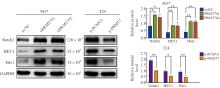
Fig. 4
Notch signaling pathway related proteins were detected PRMT7 gene was silenced in 5637 cells, and PRMT7 gene was overexpressed in T24 cells. Compared with the control group, the expression of Notch1, HEY1 and Hes1 increased after down-regulation of PRMT7, while the overexpression of PRMT7 was on the contrary. *: P<0.05, compared with si-NC or p-PCMV3; **: P<0.01, compared with si-NC or p-PCMV3."
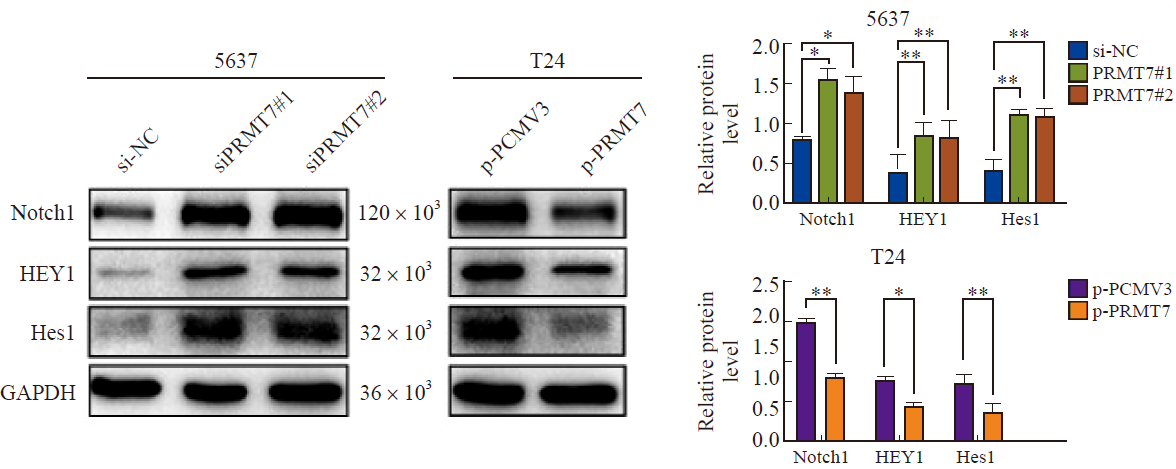
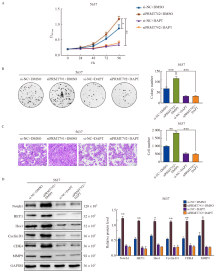
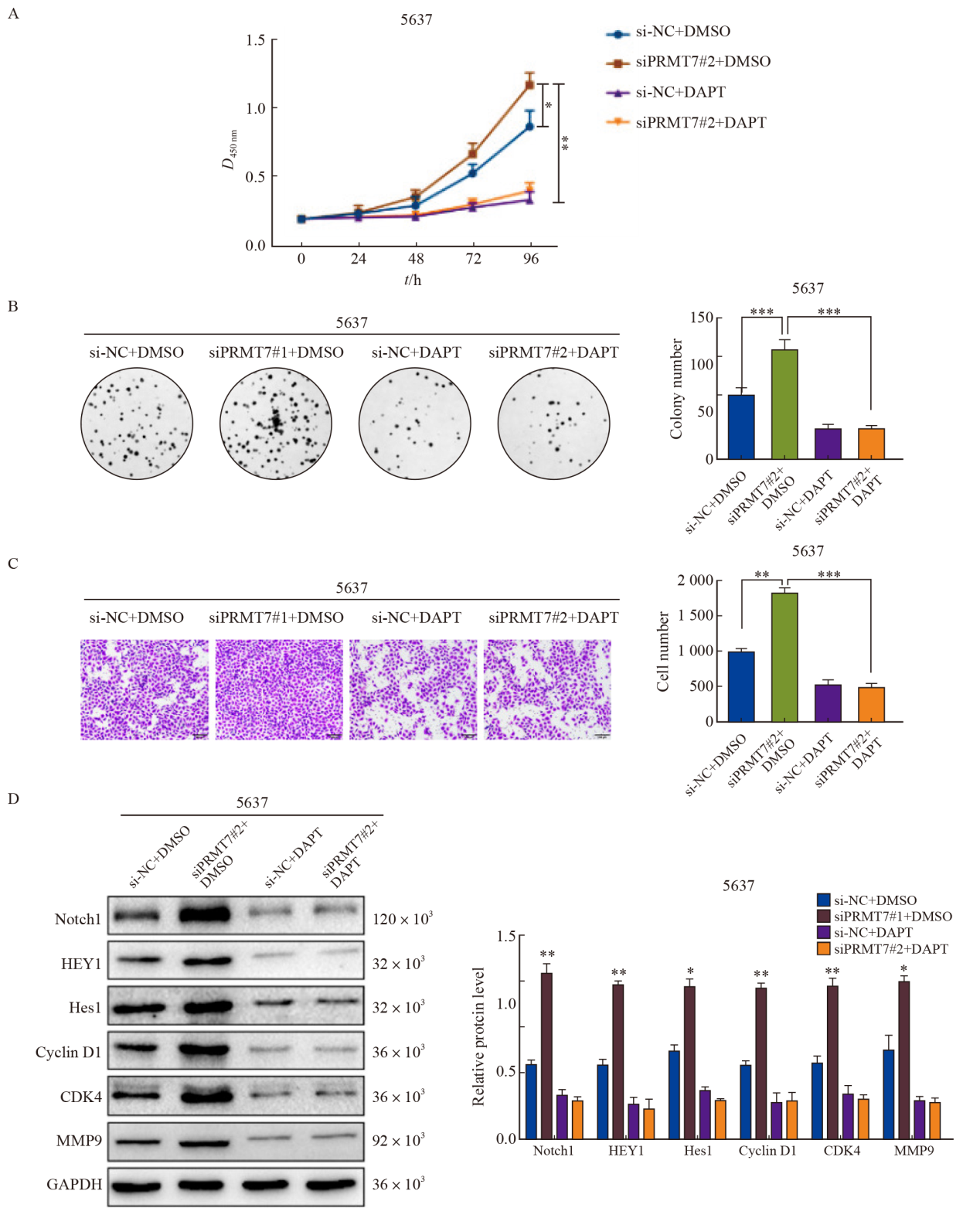
Fig. 5
PRMT7 inhibits the proliferation and migration of bladder cancer cells by regulating Notch signaling pathway 5637 cells were transfected with siPRMT7#2 and treated with DAPT. A: CCK-8 experiment showed that DAPT treatment inhibited cell growth. B: Colony formation experiment showed that DAPT treatment inhibited cell proliferation. C: Transwell experiment showed that DAPT treatment inhibited cell migration. D: Western blot results showed that compared with si-NC group, DAPT reversed the expression of Notch1, HEY1, Hes1 and the downstream proliferation and migration related target proteins Cyclin D1, CDK4, MMP9. *: P<0.05, compared with each other; **: P<0.01, compared with each other; ***: P<0.001, compared with each other."
| [1] |
SUNG H, FERLAY J, SIEGEL R L, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249.
doi: 10.3322/caac.v71.3 |
| [2] | 钟键, 金正贤, 卞卫星, 等. FGFR3和PIK3CA基因突变影响膀胱癌的预后[J]. 中国癌症杂志, 2019, 29(11): 880-886. |
| ZHONG J, JIN Z X, BIAN W X, et al. Effects of FGFR3 and PIK3CA mutations on prognosis of bladder carcinoma[J]. China Oncol, 2019, 29(11): 880-886. | |
| [3] |
LENIS A T, LEC P M, CHAMIE K, et al. Bladder cancer: a review[J]. JAMA, 2020, 324(19): 1980-1991.
doi: 10.1001/jama.2020.17598 pmid: 33201207 |
| [4] |
CHEN X, XIE R H, GU P, et al. Long noncoding RNA LBCS inhibits self-renewal and chemoresistance of bladder cancer stem cells through epigenetic silencing of SOX2[J]. Clin Cancer Res, 2019, 25(4): 1389-1403.
doi: 10.1158/1078-0432.CCR-18-1656 |
| [5] |
XU J, RICHARD S. Cellular pathways influenced by protein arginine methylation: implications for cancer[J]. Mol Cell, 2021, 81(21): 4357-4368.
doi: 10.1016/j.molcel.2021.09.011 pmid: 34619091 |
| [6] |
HE H C, CHEN J L, ZHAO J, et al. PRMT7 targets of Foxm1 controls alveolar myofibroblast proliferation and differentiation during alveologenesis[J]. Cell Death Dis, 2021, 12(9): 841.
doi: 10.1038/s41419-021-04129-1 pmid: 34497269 |
| [7] |
YAO R S, JIANG H, MA Y H, et al. PRMT7 induces epithelial-to-mesenchymal transition and promotes metastasis in breast cancer[J]. Cancer Res, 2014, 74(19): 5656-5667.
doi: 10.1158/0008-5472.CAN-14-0800 pmid: 25136067 |
| [8] |
ASTER J C, PEAR W S, BLACKLOW S C. The varied roles of Notch in cancer[J]. Annu Rev Pathol, 2017, 12: 245-275.
doi: 10.1146/annurev-pathol-052016-100127 pmid: 27959635 |
| [9] |
LI W J, HE Y H, YANG J J, et al. Profiling PRMT methylome reveals roles of hnRNPA1 arginine methylation in RNA splicing and cell growth[J]. Nat Commun, 2021, 12(1): 1946.
doi: 10.1038/s41467-021-21963-1 |
| [10] |
JEONG H J, LEE S J, LEE H J, et al. PRMT7 promotes myoblast differentiation via methylation of p38MAPK on arginine residue 70[J]. Cell Death Differ, 2020, 27(2): 573-586.
doi: 10.1038/s41418-019-0373-y |
| [11] |
CHEN M, WANG Y B, LIN L M, et al. PRMT7 is involved in regulation of germ cell proliferation during embryonic stage[J]. Biochem Biophys Res Commun, 2020, 533(4): 938-944.
doi: 10.1016/j.bbrc.2020.09.099 |
| [12] |
LIU L X, ZHANG X L, DING H Y, et al. Arginine and lysine methylation of MRPS23 promotes breast cancer metastasis through regulating OXPHOS[J]. Oncogene, 2021, 40(20): 3548-3563.
doi: 10.1038/s41388-021-01785-7 pmid: 33927350 |
| [13] |
SHEN T, NI T, CHEN J X, et al. An enhancer variant at 16q22.1 predisposes to hepatocellular carcinoma via regulating PRMT7 expression[J]. Nat Commun, 2022, 13(1): 1232.
doi: 10.1038/s41467-022-28861-0 pmid: 35264579 |
| [14] |
ROPER N, VELEZ M J, CHIAPPORI A, et al. Notch signaling and efficacy of PD-1/PD-L1 blockade in relapsed small cell lung cancer[J]. Nat Commun, 2021, 12(1): 3880.
doi: 10.1038/s41467-021-24164-y pmid: 34162872 |
| [15] |
GENG Y W, FAN J, CHEN L Y, et al. A Notch-dependent inflammatory feedback circuit between macrophages and cancer cells regulates pancreatic cancer metastasis[J]. Cancer Res, 2021, 81(1): 64-76.
doi: 10.1158/0008-5472.CAN-20-0256 pmid: 33172931 |
| [16] |
HE H D, SHAO X N, LI Y N, et al. Targeting signaling pathway networks in several malignant tumors: progresses and challenges[J]. Front Pharmacol, 2021, 12: 675675.
doi: 10.3389/fphar.2021.675675 |
| [17] | ZHOU B H, LIN W L, LONG Y L, et al. Notch signaling pathway: architecture, disease, and therapeutics[J]. Signal Transduct Target Ther, 2022, 7(1): 95. |
| [18] |
HARADA Y, YAMADA M, IMAYOSHI I, et al. Cell cycle arrest determines adult neural stem cell ontogeny by an embryonic Notch-nonoscillatory Hey1 module[J]. Nat Commun, 2021, 12(1): 6562.
doi: 10.1038/s41467-021-26605-0 pmid: 34772946 |
| [19] |
ZHU J J, LI X, CAI X L, et al. Arginine monomethylation by PRMT7 controls MAVS-mediated antiviral innate immunity[J]. Mol Cell, 2021, 81(15): 3171-3186.e8.
doi: 10.1016/j.molcel.2021.06.004 |
| [1] | WEN Ziqiang, LAN Junliang, ZHOU Bo, XU Qiwei. PARP1 promotes the progression of hepatocellular carcinoma by regulating expression of POU2F2 [J]. China Oncology, 2024, 34(9): 848-856. |
| [2] | CAO Fei, YU Wenhao, TANG Xiaonan, MA Zidong, CHANG Tingmin, GONG Yabin, LIAO Mingjuan, KANG Xiaohong. Mechanism of LINC01410 promoting proliferation and migration in esophageal squamous cell carcinoma [J]. China Oncology, 2024, 34(8): 753-762. |
| [3] | CHEN Xun, ZHENG Zhenxia, RUAN Xueru. Effects of TMCO1 on proliferation and migration of cervical cancer cells [J]. China Oncology, 2024, 34(6): 571-580. |
| [4] | Urologic Chinese Oncology Group. Expert consensus on early diagnosis and treatment of bladder cancer (2024 edition) [J]. China Oncology, 2024, 34(6): 607-618. |
| [5] | SUN Rongqi, SONG Ning, ZHENG Wentian, ZHANG Xinyue, LI Minmin, GONG Hui, JIANG Yingying. Effect of long noncoding RNA FLJ30679 on proliferation and migration of oral squamous cell carcinoma cells [J]. China Oncology, 2024, 34(5): 439-450. |
| [6] | XIONG Jiayan, LEI Wei, YOU Bo, ZHANG Zhenxin, XIE Haijing, SHAN Ying, XIA Tian, ZHOU Yong. Study on the mechanism of DDX6 promoting proliferation and migration of nasopharyngeal carcinoma cells by regulating stability of CKMT1A mRNA [J]. China Oncology, 2024, 34(5): 451-459. |
| [7] | ZHOU Xueqin, LUAN Yanchao, ZHAO Li, RONG Chaochao, YANG Na. Expression of CDC20 in lung adenocarcinoma tissues and its effect on the proliferation and invasion of lung adenocarcinoma cells [J]. China Oncology, 2024, 34(5): 460-472. |
| [8] | GUAN Ruirui, HAO Qian, ZHANG Yaqi, SUN Qinggang, CHEN Yitian, LI Xiumin, ZHOU Xiang, HAN Tao. CDC20 facilitates the proliferation of esophageal carcinoma cell by stabilizing NLRP3 expression [J]. China Oncology, 2024, 34(5): 473-484. |
| [9] | XU Ziqi, HU Ruizhi, LI Junjian, WANG Hongxia, SANG Youzhou. Exploring the role of methylation-driven gene IFFO1 in pancreatic adenocarcinoma diagnosis, prognosis and cellular functions [J]. China Oncology, 2024, 34(11): 998-1010. |
| [10] | JIA Liqing, GE Xiaolu, JIANG Lin, ZHOU Xiaoyan. Effects of lncRNA PKD2-2-3 on cell proliferation, clone formation, migration, and invasion of lung adenocarcinoma [J]. China Oncology, 2023, 33(8): 717-725. |
| [11] | ZHANG Pingchuan, DU Mingyu, YAO Chengyun, HE Xia, YIN Li. Mechanism of circular RNA hsa_circ_0012779 expression in nasopharyngeal carcinoma and its influence on cell biological behavior [J]. China Oncology, 2023, 33(5): 445-451. |
| [12] | TANG Duocai, ZHOU Shukui, ZHANG Guiyin, LIU Lei, LIAO Hong. Analysis of risk factors of recurrence in non-muscle invasive bladder cancer patients after initial transurethral resection of bladder tumor [J]. China Oncology, 2023, 33(5): 478-483. |
| [13] | XIAO Lanshu, PAN Liudi, LIU Yi, WANG Jie, CHEN Hui. LncRNA DLEU7-AS1 contributes to proliferation and migration of gastric cancer by regulating MSN transcription [J]. China Oncology, 2023, 33(4): 327-341. |
| [14] | CHEN Hong, CHEN Junxia. Effect of hsa_circ_0001573 on biological behaviors of breast cancer cells and its molecular mechanism [J]. China Oncology, 2023, 33(4): 342-353. |
| [15] | PENG Jin, WANG Weining, TAN Zhi, YE Guannan, ZHOU Zhen. The mechanism of m6Am-modifying enzyme PCIF1 regulating target gene ACOT8 in gastric cancer progression [J]. China Oncology, 2023, 33(4): 368-376. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
沪ICP备12009617
Powered by Beijing Magtech Co. Ltd
